The Chemistry of Mitragynine & 7-Hydroxymitragynine: Unlocking Alkaloid Pharmacology

When you are actively evaluating botanical options, you quickly outgrow marketing buzzwords. If you want to accurately predict how a specific batch will behave—whether you are comparing a standard leaf or a potent kratom extract powder—you need to look under the hood. You need to understand the chemistry.
At the heart of the Mitragyna speciosa tree are two primary alkaloids: Mitragynine (MG) and 7-Hydroxymitragynine (7-OH). By translating the dense pharmacological data surrounding these compounds into practical insights, you can bridge the gap between raw science and your actual experience.
Here is the foundational chemical knowledge you need to evaluate products with confidence.
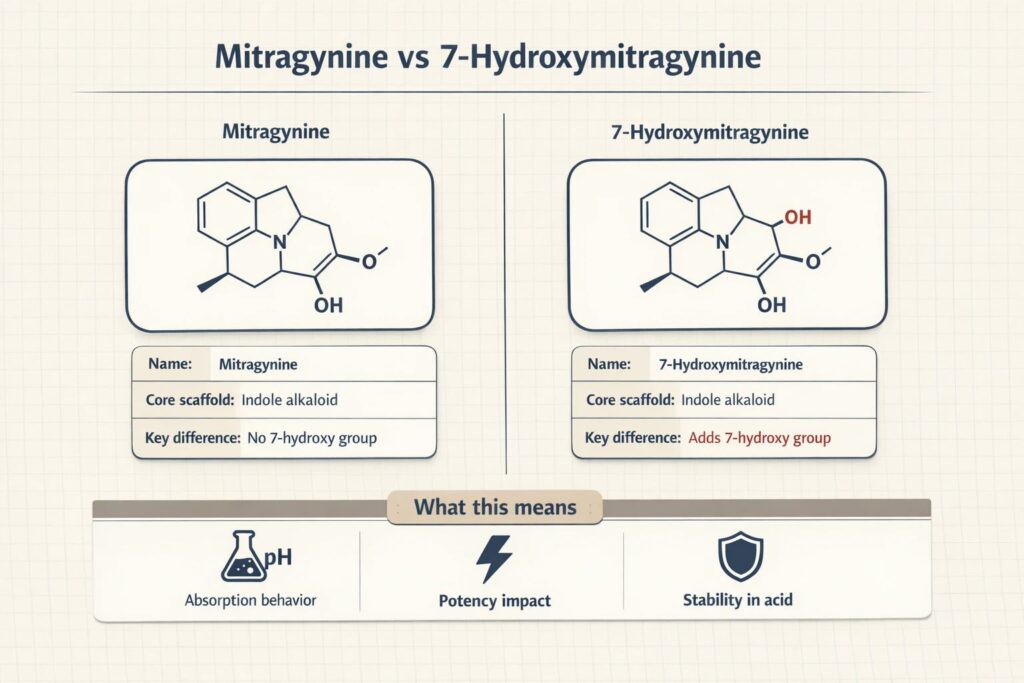
The Molecular Blueprint and Biosynthesis
To understand how these alkaloids work, we have to look at how the plant builds them. The journey begins with the Shikimic acid pathway. In the leaves of the kratom tree, this biochemical route synthesizes the amino acid tryptophan, which ultimately provides the essential indole alkaloid core found in both MG and 7-OH.
Mitragynine makes up roughly 66% of the total alkaloid content in typical leaf matter. Its molecular structure features this robust indole core, heavily influencing how the body metabolizes it. But nature rarely stops at a single compound. Through natural oxidation and metabolic processes, the plant also produces 7-Hydroxymitragynine.
While the two share nearly identical frameworks, a minor functional-group change at the molecular level drastically alters how they bind to receptors.
Physicochemical Properties: The Numbers That Dictate Bioavailability
If you have ever weighed the pros and cons of kratom powder vs extract, you are fundamentally evaluating physicochemical properties. How a compound breaks down and absorbs into the bloodstream relies entirely on its solubility, pH sensitivity, and partition coefficient.
pKa and LogP Variables
According to established peer-reviewed models, Mitragynine has a pKa value of 8.11 and a LogP of 1.73.
- LogP (Lipophilicity): A LogP of 1.73 indicates that MG is moderately lipophilic (fat-soluble). It can cross cell membranes efficiently, which explains its relatively rapid onset when absorbed.
- pKa (Ionization): The pKa of 8.11 means that in basic environments, MG remains largely un-ionized and lipid-soluble, but in highly acidic environments, it becomes ionized and water-soluble.
The Acid Labile Factor
This is where the environment of the human stomach comes into play. Research utilizing Simulated Gastric Fluid (SGF) at a highly acidic pH of 1.2 reveals a critical vulnerability: Mitragynine loses greater than 20% of its active content within just 20 minutes of exposure.
Conversely, when tested in Simulated Intestinal Fluid (SIF), which has a more neutral pH, the alkaloid remains highly stable. Furthermore, testing shows MG achieves an optimal solubility of 3.5 mg/mL at a mildly acidic pH of 4. This acid degradation factor is a primary reason why formulation, extraction method, and consumption technique drastically impact the final biological availability of the alkaloids.

The “Potency Switch” and Diastereomers
Why does a product with a nearly identical milligram weight feel completely different from another? The answer lies in the “Potency Switch”—the presence of 7-Hydroxymitragynine and other minor alkaloids.
7-OH: The Minor Alkaloid with Major Impact
7-OH naturally occurs in very low concentrations, typically making up less than 2% of the total alkaloid profile in raw leaf material. However, pharmacological data shows that 7-OH is approximately 46 times more potent than Mitragynine and up to 13 times more potent than traditional morphine at specific receptor sites.
When consumers upgrade to an enhanced kratom powder, they are essentially paying for a stabilized, concentrated ratio of these alkaloids, capturing that amplified potency that raw leaf alone struggles to provide.
Diastereomers: A Simple Inversion
The chemistry gets even more fascinating when looking at diastereomers like Speciociliatine. By simply inverting a single hydrogen atom at the C-3 position of the molecular structure, the alkaloid’s behavior fundamentally shifts. A compound can change from acting as a partial agonist to exhibiting potential antagonist properties.
This molecular variability is the true scientific basis behind any kratom strain chart. When you read about the differences between “vein colors,” you are actually reading about varying ratios of MG, 7-OH, and these crucial diastereomers resulting from different drying and oxidation processes.
Pharmacology Bridge: Contextualizing Kratom
To fully evaluate these chemical profiles, it helps to see how they map against broader pharmacological standards.
Where Does it Fit in the 6 Classifications of Drugs?
Educational and regulatory frameworks generally divide substances into six classifications: Stimulants, Depressants, Hallucinogens, Opioids, Inhalants, and Cannabis. Kratom possesses a highly atypical profile that straddles categories. At lower concentrations, its alkaloid profile exhibits stimulant-like effects. At higher concentrations, the 7-OH dominance binds more heavily to mu-opioid receptors, resulting in opioid-like analgesic behavior. It is this dose-dependent dualism that makes the plant’s chemistry so unique.
The ADH Connection
Alkaloids don’t just interact with pain or energy pathways; they interact with the endocrine system. The pituitary-adrenal axis controls the release of Antidiuretic Hormone (ADH), which dictates how your body retains water. Because of how indole alkaloids interact with autonomic pathways, heavy or concentrated usage can occasionally impact ADH regulation, underscoring why proper hydration is frequently recommended when exploring deeper pharmacological dosages.

Chemistry FAQ
How do kratom alkaloids dissolve compared to over-the-counter tablets?
A common user query compares dissolving kratom to dropping a standard Disprin (aspirin) tablet in water. Disprin is formulated with effervescent agents to rapidly lower pH and achieve immediate water solubility. Kratom alkaloids, in their natural state, are hydrophobic (water-repelling). Without the addition of an acidic medium (like citric acid from lemon juice) to lower the pH and ionize the alkaloids, they will suspend in water rather than fully dissolve.
Is chlorine used to make kratom extracts?
When evaluating products, users sometimes see the term “Hydrochloride” (HCl) and mistakenly worry about toxic chlorine gas. In chemistry, reacting an alkaloid base with hydrochloric acid is a standard, safe pharmaceutical practice used to create a “salt.” This process simply stabilizes the molecule and massively increases its water solubility, which improves absorption. It leaves no toxic chlorine behind, only a highly stable, bioavailable compound.
Confident Decision Making
When evaluating your options, raw botanical data is your greatest asset. Understanding that Mitragynine degrades in high acid while 7-OH dictates the potency ceiling allows you to make precise, informed decisions about which formats suit your specific routine. Whether you are looking for the rapid onset of an extracted salt or the complex, full-spectrum profile of natural leaf, leaning on the chemistry ensures you know exactly what you are putting into your body.